|
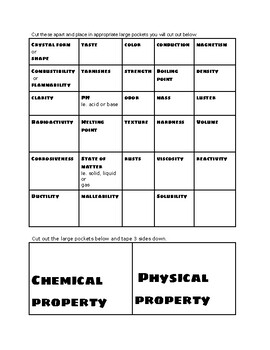
The formation of gas bubbles is often the result of a chemical change (except in the case of boiling, which is a physical change). For example, the freezing point of a substance is a physical property: when water freezes, it’s still water (\text Both extensive and intensive properties are physical properties, which means they can be measured without changing the substance’s chemical identity. Intensive properties, such as density and color, do not depend on the amount of matter. Extensive properties, such as mass and volume, depend on the amount of matter that is being measured. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
